MXene researchers find 2-D transition metal carbides react with water, opening a door to their unknown chemistry
Posted by Delia Croessmann
On January 16, 2019
Researchers at Missouri University of Science and Technology have discovered that 2-D titanium carbide materials, or MXenes, can react with water without the presence of other oxidizers. Their finding may lead to new insights into the unusual chemistry of MXenes and consequently, have impacts on MXenes’ storage and device manufacturing.
“Our new findings are important because now we know it is water itself rather than oxygen that MXenes need to be protected from during manufacturing and storage,” says Shuohan Huang, a doctoral student in chemistry at Missouri S&T.
MXenes are one of the largest families of 2-D materials. They are a few atom-thick sheets with the structures of transition-metal carbides and nitrides. Their distinctive properties include high electrical conductivity and the ability to disperse in water, a unique combination which earned them a nickname “conductive clays.”
Researchers are excited about the potential use of MXenes in energy storage and harvesting applications such as batteries, supercapacitors and triboelectric nanogenerators, which convert wasted frictional energy into electricity, according to Dr. Vadym Mochalin, associate professor of chemistry at Missouri S&T, who is the principal investigator of this project.
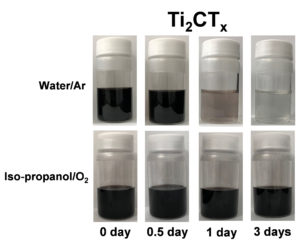
MXenes’ reactivity toward water is shown in this experiment where a Ti carbide MXene survives longer in an oxygen-saturated non-aqueous system, in contrast to the same MXene dispersed in argon-saturated water. Photo credit: Shuohan Huang.
“The reactivity of MXenes toward water we’ve demonstrated not only changes the common perception about resistance of titanium carbide to hydrolysis in ambient conditions, but also points out the striking differences in chemical properties between bulk and 2-D forms of the same material,” says Mochalin.
Mochalin and lead author Huang, who works in Mochalin’s research group at Missouri S&T, discuss their findings in a paper titled “Hydrolysis of 2D Transition Metal Carbides (MXenes) in Colloidal Solutions,” which was published today (January 16, 2019) in the American Chemical Society journal Inorganic Chemistry.
To arrive at their “water-only” conclusion, Huang and Mochalin systematically tested the hydrolysis and chemical stability of MXenes Ti3C2Tx and Ti2CTx in water and non-aqueous colloidal solutions exposed to oxygen and/or inert gas environments in different combinations, as the example shown in the image.
“It seems that in their 2-D state, transition-metal carbides are quite reactive,” says Mochalin. “With our result, we’re looking forward to follow-up studies of their rich chemistry in reactions with water and other molecules, including organic compounds, as well as studies into MXenes’ possible catalytic properties.”
Share this page
Posted by Delia Croessmann
On January 16, 2019. Posted in Academics & Faculty, CASB Research, College of Arts, Sciences, and Business, Department of Chemistry, Missouri S&T, Missouri University of Science and Technology, Research, Science & Tech, Science Etc.
Looking for something?
Recent Posts
- Missouri S&T honors Patrick Wagner for professional distinction
- Gibbs talks of shared experience with S&T grads
- Brewer shares the important role of failure in success with S&T grads
- From the lab to the fire line: Two S&T students chase the heat
- S&T researcher selected for national fellowship on societal impact
Recent Comments
- Matthew Wells on Missouri governor to host artificial intelligence and data center forum at Missouri S&T
- Hugh V STEWART on Missouri S&T professor elected to American Concrete Institute board
- Jill Hollowell on Missouri governor to host artificial intelligence and data center forum at Missouri S&T
- Dan Mehan on Missouri governor to host artificial intelligence and data center forum at Missouri S&T
- John Doyle on Missouri S&T alumnus Lister Florence takes the reins as Rolla mayor
Upcoming events
Missouri S&T is an equal opportunity employer
© 2026 - Curators of the University of Missouri | WordPress
Leave a Reply